Evaporative sealants
When an evaporative curing sealant I believe is what it’s called, dap extreme stretch (not a chemically curing sealant) is closed up but not sandwiched between two impervious surfaces, will it still cure? I have a situation where either I need to wait two weeks for this to fully cure in the open air or close it up prior to full cure, which would be ideal. One side of the sandwich so to speak would be impervious, the other side being wood.
Dap says this can be painted in two hours but the tech support also advised it takes two weeks to cure, so I’m assuming it’s ok to cover up at least after the two hours… I’ve never really understood this concept
Thanks for any help
GBA Detail Library
A collection of one thousand construction details organized by climate and house part
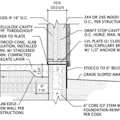
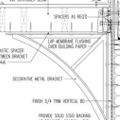






Replies
What is "impervious"? Is it metal foil of some kind, something like that? Metal and glass are about the only normal materials that can really prevent proper curing here.
What you're referring to is known as a "solvent cure" sealant, which means the sealant cures when the solvent evaporates out. Silicone, for example, uses acetone as a solvent (which is where the smell comes from), and when the acetone evaporates away, you get the usual semisolid silicon instead of the more liquid silicone you squeezed out of the tube. Acetone also can clean up silicone messes pretty well.
The nice thing about those solvents is they will usually go through things like poly sheeting if you have any of that in a wall. There is a reason why the inside of those caulk tubes are metalized :-) In your assembly, you'll probably be OK, but cure time might be a little longer than expected. Solvent cure materials in confined spaces will sometimes cure more slowly since the higher concentration of evaporarted solvent in the confined space slows down the evaporation of the remaining solvent in the product.
If you are concerned, test a sample and see if it's fully cured after a few hours. If it is, you're good to go.
Bill